Medical Device Innovation Consortium Case for Quality Forum
On November 6th, CEO Jack Garvey will be attending the MDIC Case for Quality Forum in Washington, D.C. The Forum will focus the Case for Quality Voluntary Improvement Program (CFQ VIP) and plans to grow the program in 2020 and beyond. Topics include: A keynote presentation on Collaborations in Digital Health and new initiatives at […]
CDRH Proposed Guidances for Fiscal Year 2020 (FY 2020)
from FDA The lists on this page include guidance documents that CDRH intends to publish this fiscal year (FY 2020) and previously issued final guidances for which CDRH is interested in receiving external feedback regarding whether these final guidances should be revised or withdrawn. These lists are: A-List: Prioritized Guidance Documents that CDRH Intends to […]
CEO Jack Garvey to Co-Host FDLI Webinar “Due Diligence: Ensuring Your Deal Doesn’t Collapse Due to Regulatory, Clinical, Quality, or Data Integrity Issues”
With the increasing difficulties in internally-generated product development, drug, biologics, and device firms of all sizes are looking to acquisitions to augment product lines. Whether in the form of single products or whole companies, transactions can make or break the future of a company. However, often lost in the frenzy of a deal involving highly-regulated […]
CEO Jack Garvey to Present at Association for GXP Excellence Spring Meeting 2019
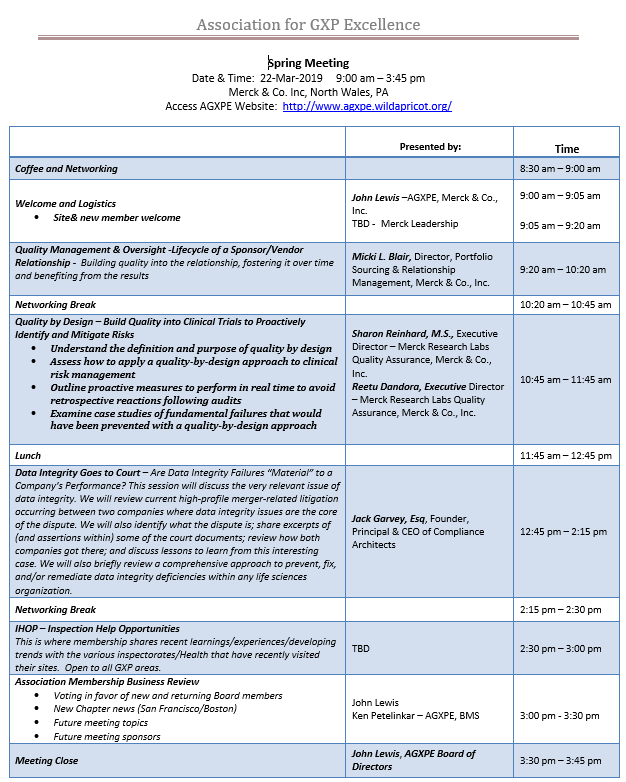
Our CEO Jack Garvey is very excited to be presenting today at AGXPE’s 2019 Spring Conference this afternoon. More about the meeting: “This meeting is focused on vendor quality and oversight with and End-to-End perspective. Merck’s team of volunteer presenters never seem to disappoint the audience as they share key learnings and insights. Like always, […]
FDA Xavier PharmaLink 2019 in Cincinnati, Ohio

We’re excited to be attending the FDA/Xavier PharmaLink in Cincinnati, Ohio. This marks our first visit to the conference, and we’re eager to hear what some of the brightest minds in the industry have to say. Every attendee will be going home with one of our brand-new Writing for Compliance® pads! The conference seeks solutions […]
Jeff Grizzel Joins Compliance Architects LLC as Vice President, Business Development
Compliance Architects® announced today the appointment of Jeff Grizzel to the company’s leadership as Vice President, Business Development. Grizzel’s appointment bolsters the company’s focus to deliver world-class compliance and quality consulting, outsourcing, staff augmentation and technology-related services to FDA-regulated companies worldwide. Compliance Architects® continues to innovate and exceed client expectations through an expanded portfolio of […]
Jack Garvey to lead Quality Stream discussion at 2018 Biomanfacturing Wold Summit
Compliance Architects® is excited to announce that its Chief Executive Officer, Jack Garvey, will be the chairperson for the Quality Stream on October 29th at the 2018 Biomanufacturing World Summit in San Diego, California. As the global biopharmaceutical industry continues to grow and evolve, companies are constantly facing pressures to focus on the patient as they […]
Compliance Architects® CEO Jack Garvey to Co-Present with Bayer Pharmaceutical VP of Quality Tina Self on Strategic Quality Plan Development

Compliance Architects® is excited to announce that its Chief Executive Officer, Jack Garvey, will co-present on the topic of Strategic Quality Plan Development with Bayer Pharmaceutical, Berkeley Site, Vice President of Quality, Tina Self. The presentation, to be delivered at the Generis American Biomanufacturing Summit, 2018, in San Francisco, CA on June 14 and 15th, […]
Compliance Architects® is Bronze Sponsor of 2018 FDLI Annual Conference
Once again, Compliance Architects® is proud to announce that it will be a Bronze Sponsor of the FDLI Annual Conference. This year’s event, to be held in Washington, DC on May 3 & 4, 2018, is a must-attend event for legal, regulatory and compliance executives within FDA-regulated industry. The Food and Drug Law Institute (FDLI) is the country’s premiere organization dedicated to […]
Jack Garvey Cited for Investment Community Research on Data Integrity
Reflecting Compliance Architects® strong, demonstrated expertise in data integrity risk identification and resolution within the pharmaceutical and related industries, Jack Garvey, Founder and Chief Executive Officer of Compliance Architects® was cited recently by Event Driven, an investment news and analysis subscription product of Reorg Research. Jack provided insight and analysis to Event Driven on the impact […]

